-
![]()
The Hamilton Spectator ‘Some teachers leave a mark that lasts a lifetime’ February 17, 2026
New book sent by a former student to a retired teacher exposes the lax governance of medical research that puts at risk the safety and health of human research participants.
-
![]()
CBC News Article - December 04, 2024
'No consequences' for violating human rights in privately funded research in Canada, says ethics expert. Ethics experts say Canadians should think twice about becoming research participants, because the federal body that holds universities accountable to ethical standards has no jurisdiction over privately funded studies.
-
![]()
Pharma Boardroom Interview - December 19, 2017
“Canada could find advantage by positioning itself as a country of excellence for smaller studies or more complex studies requiring innovative designs, the type ethica CRO has excelled with in the past."
-
![]()
Women of Influence Magazine Article - Fall 2014
Trials & Errors: Life saving drugs must be tested on human subjects before they’re made available to us all. One woman is pushing hard to make sure those subjects get the humanity they deserve.
-
![]()
Women of Influence Magazine Interview - October 23, 2014
Dr. Janice Parente: A scientist with entrepreneurial DNA
-
![]()
More Magazine Article - Summer 2007
Guarding the Guinea Pigs: Human volunteers in clinical drug trials are less protected than animals. One woman is working to change that.
-
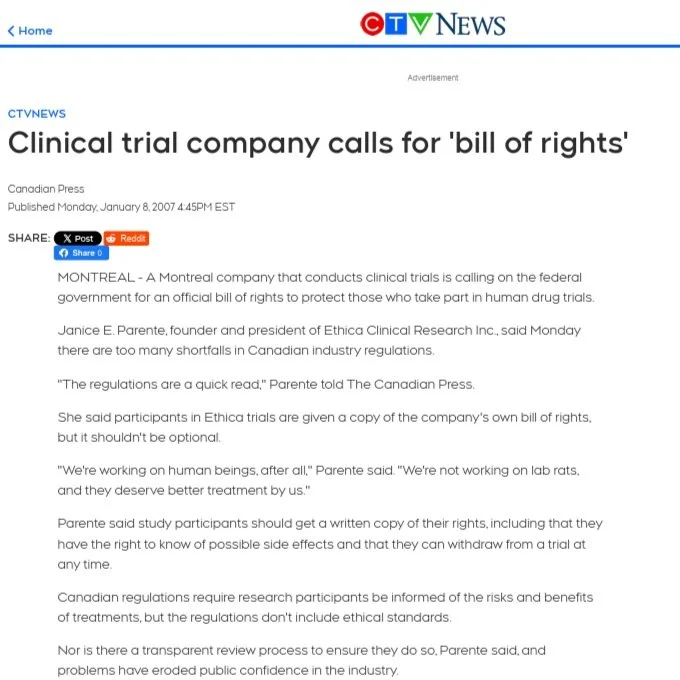
![]()
CTV News Article - January 08, 2007
Clinical trial company calls for 'bill of rights'
-
![]()
CBC News Article - January 08, 2007
Human test subjects need protection - A Montreal company that conducts clinical trials is calling on the federal government for an official bill of rights to protect those who take part in human drug trials.